The UK’s premier cosmetic group
Specialists in cosmetic procedures
Procedure areas
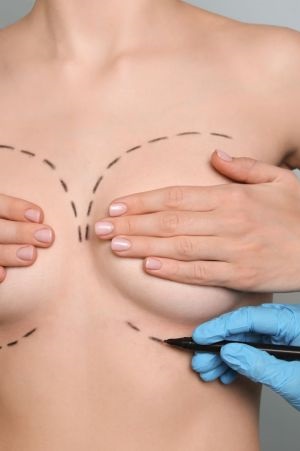
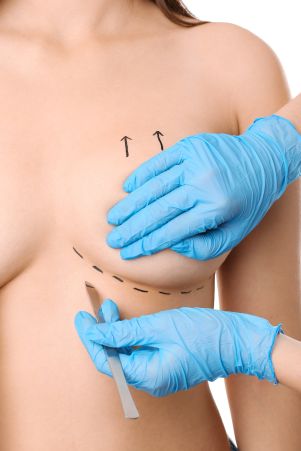
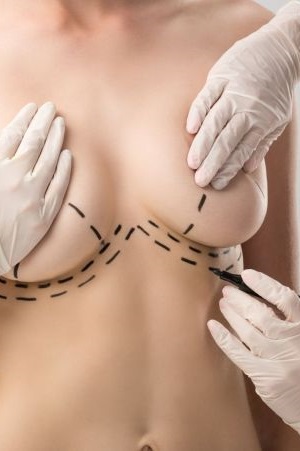
The team at MyBreast pride ourselves in being specialists in all areas of cosmetic surgery from breast enlargement and tummy tucks to fat transfer and facial surgery. We ensure the highest level of patient care from the moment of your initial enquiry right until you have fully recovered and enjoying your new you.
Locations
Your care will take place at one of two 5* private facilities. The Weymouth Street Hospital is located in the world renowned Harley Street District in Central London with its sister Hospital, Phoenix Hospital Chelmsford located in Essex
Both locations offer not only exceptional care and service, they are also committed to our patients, continually striving to deliver excellence in all aspects of your journey.
Testimonials
![]()
"The quality of service received at MyBreast was exceptional. My surgeon was professional, warm and provided a clear outlook in terms of surgery objectives and outcomes right from the start. Assisting me throughout this journey my patient advisor was very knowledgeable; always providing maximum support and guidance. I am very thankful to everyone who was involved and I am extremely pleased with the end result of my surgery, as well as the journey that has taken me there."
Sourced from MyBreast feedback survey
![]()
"I am absolutely delighted with my Breast reduction/ Mastoplexy. I would recommend “My breast” to anyone who is considering having breast surgery of any type. My surgeon, Professor Sforza was amazing and I couldn’t recommend him highly enough. The whole team worked together and delivered a five star service. Thank you, you’ve made my life so much better."
Sourced from MyBreast feedback survey
![]()
"Was looked after so so well, the nurses were extremely kind and polite, and catered to my every need. The procedure was quick and I felt no pain! I was clearly in the hands of very talented people and I also felt very safe and well looked after. I would highly recommend MyBreast to anyone who wanted to boost their confidence through cosmetic surgery!"
Sourced from TrustPilot
Get in touch
Look no further for your dream body.
Get in touch today to speak to one of our friendly advisors who will be able to book you in for an initial consultation.
9 Harley St | London | W1G 9QY